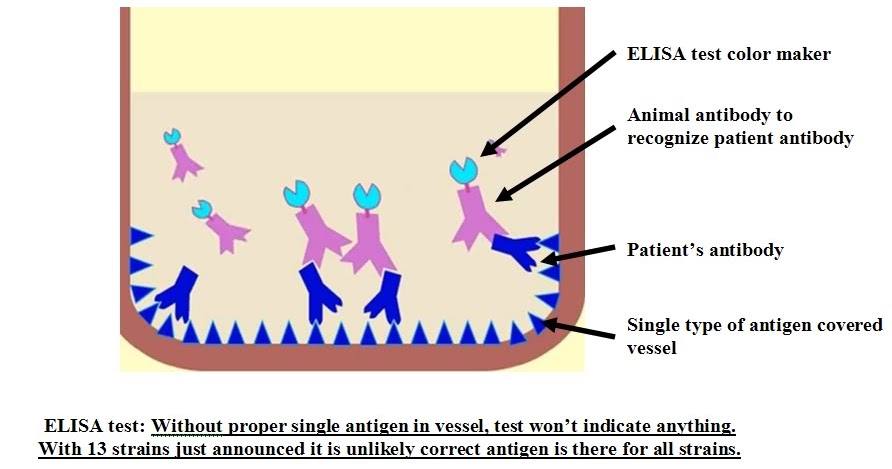
Why is an ELISA test so sensitive?ĮLISAs tend to be the most sensitive immunoassays due to the binding characteristics of the antibodies and the amplification or different read-out systems used. The potential of observing cross-reactivity or interference is minimized and you can push the sensitivity limits. An advantage of ELISAs over different multiplexing platforms is the ability to customize the assay for that antigen and not having to worry about many other antibodies and proteins working together. Comparing an ELISA to a Western blot, sandwich ELISAs use 2 specific antibodies rather than one and allow for completely quantitative results, while a Western blot can see non-specific bands and are semi-quantitative at best. ELISAs are preferred in many cases due to their sensitivity, specificity, accuracy, and ability to tolerate harsh buffers or pretreatments. There are many different immunoassay platforms available to measure to quantitate protein levels in biological fluids. An advantage to using indirect detection is that you can use the same secondary antibody for multiple different assays, eliminating the need to conjugate primary antibodies for every ELISA. Indirect ELISAs also take longer due to the extra step. Indirect ELISAs can be more sensitive than direct ELISA detection methods due to the amplification step, but there is also the risk of cross-reactivity with the antigen, which could cause higher background. In an indirect ELISA, an unconjugated primary antibody binds to the antigen, then a labeled secondary antibody directed against the host species of the primary antibody binds to the primary antibody. Direct ELISAs use a conjugated primary antibody, while indirect ELISAs include an additional amplification step. The difference in a direct vs indirect ELISA is in the detection method of the immobilized antigen on an ELISA plate. Investigating blocking/inhibitory interactions. When to Use: Assessing antibody affinity and specificity. Since only one antibody is used in a direct ELISA, they are less specific than a sandwich ELISA. Substrate is then added, producing a signal that is proportional to the amount of analyte in the sample. In a direct ELISA, an antigen or sample is immobilized directly on the plate and a conjugated detection antibody binds to the target protein. Each type of ELISA has its own advantages and disadvantages. The four main types of ELISAs are direct, indirect, sandwich, and competitive. A substrate solution is subsequently added to produce a signal that is proportional to the amount of analyte bound. A conjugated detection antibody that binds to a different epitope on the target analyte is used to complete the sandwich. Upon incubation with experimental samples, standards, or controls, the target analyte is captured by this antibody. Samples routinely used in ELISAs include serum, plasma, cell culture supernates, cell lysates, saliva, tissue lysates, and urine.ĮLISAs are usually run in 96-well microplates coated with a capture antibody specific for the analyte of interest. ELISAs are a type of immunoassay that are commonly used to quantify levels of a specific target within a sample.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
